Lobular Breast Cancer: Section 5.b.
CONTENTS:
Forward to 5C on papillary types Back to 5A ductal cancer types.
5.3 Invasive Lobular Carcinoma (ILC)
Invasive lobular breast cancer (ILC) is the second most common type of invasive breast cancer, representing between 5% to 10% of all breast cancers.
With the implementation of the breast screening program, rates of detection of invasive lobular breast cancer have increased by 65% from 1987 to 1999.
In comparison, rates of ductal carcinoma have increased by only 3% (Li et al., 2003). This shows that, for some reason, invasive lobular breast cancer is on the rise.
Until recently, specialists did not histologically grade invasive lobular breast cancer. Indeed, specialists assume ILC to be a moderately-differentiated or Grade 2 carcinoma.
However, the 2012 tumor grading of invasive lobular cancer (ILC) and Pathologists will use grading systems such as the ‘Nottingham grading system’ (Elston & Ellis, 1991). The majority (75%) of classic ILC are Grade 2 (using the Nottingham grading system) with ILC Grade 3 more likely to be of solid and pleomorphic sub-type.
The 2012 WHO guidelines recommend the use of immunohistochemistry staining for E-cadherin in distinguishing ILC from NST carcinomas. However, caution is necessary because up to 15 % of ILC also express E-cadherin (e.g. the tubular-lobular variant).
Figure 5.10 Invasive Lobular Carcinoma (ILC)
Mammographic X-ray findings of ILC seen in A. the cranio-caudal
view (CC) and B. the lateral view, shows indistinct margins of the
breast mass.
There are some important features of invasive lobular carcinoma (ILC), particularly when compared with invasive ductal carcinoma (IDC) of the breast (Rakha et al., 2010).
Features of Invasive Lobular Breast Cancer
- In the U.S., the incidence of the diagnosis of lobular breast cancer is increasing more rapidly than that of ductal cancer.
- In postmenopausal women, hormone replacement therapy (HRT) may carry a stronger risk for lobular cancer than for ductal cancer risk.
- Invasive lobular carcinoma (ILC) arises in older women; the tumors tend to be larger but better-differentiated tumors
- Invasive lobular cancer has a less well-defined margin than invasive ductal carcinoma, so may be more difficult to completely excise.
- Invasive lobular carcinomas do not always present with a palpable mass. This is because a fibrous tissue reaction is less common in cases of ILC.
More Features of Invasive Lobular Breast Cancer
- Invasive lobular carcinoma (ILC) has a higher frequency of bi-laterality and multi-centricity than infiltrating ductal carcinoma (IDC).
- The microscopic appearance of ILC commonly shows small cells invading the breast in small groups or as single cells, often with ‘target-like’ arrangements around breast ducts.
- Lobular carcinoma in-situ (LCIS) is present in approximately two-thirds of cases, but DCIS may also accompany invasive lobular carcinoma.
- Invasive lobular carcinomas (ILC) are more likely than ductal carcinomas to be hormone receptor positive; usually they are estrogen (ER)-positive, with variant lesions showing occasional variable expression.
- Recent reports now show that the outcomes for invasive lobular carcinoma may be more favorable than for ductal carcinoma, but some less-favorable variants do exist (Orvieto et al., 2008).
- Invasive lobular carcinoma (ILC) tends to metastasize later than invasive duct carcinoma (IDC); this type of cancer can spread to sites such as the gastrointestinal tract and meninges.
- 50% of invasive lobular breast carcinoma (ILC) contain E-cadherin mutations (Section 6).
Figure 5.11 Cytological Appearance of Lobular Breast Cancer
Photomicroscopy of the fine needle aspiration cytology (FNAC) samples
from an invasive lobular carcinoma (ILC) shows round cells with eccentric
nuclei but with little pleomorphism, but note that they are non-cohesive.
A. Is a cell with a Giemsa smear stain
(dark blue cell nuclei and pale purple cytoplasm); B. Is a cell smear with a PAP stain.
(black cell nuclei and blue cytoplasm). (Giemsa & PAP x 100)
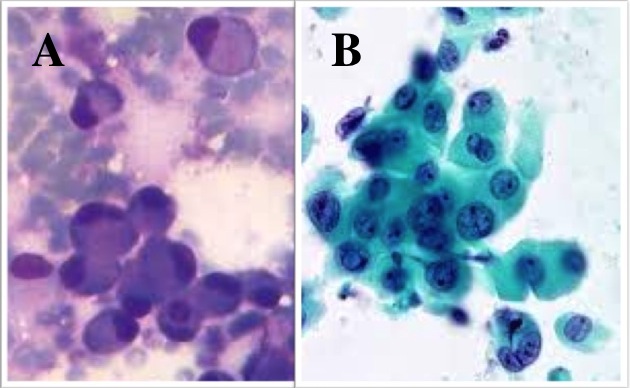
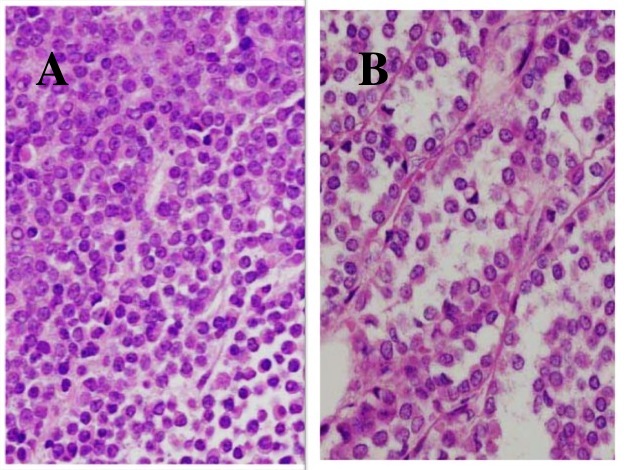
Figure 5.12 Histological Appearances of
Invasive Lobular Carcinoma
Photomicroscopy of the histology of invasive lobular carcinoma (ILC)
shows A. ‘targetoid’ infiltration by small round cancer cells, around
vessels and ducts; B. the single-cell, ‘Indian file’ invasive pattern.
(H&E x 40 & 60)
There are several variants of invasive lobular carcinoma (ILC). However, whatever the variant might be, it is the degree of differentiation or the grade that is now recognized to have prognostic meaning (Rakha & Ellis, 2010).
5.3.1 Solid Lobular Breast Cancer
The solid variant of lobular carcinoma, solid ILC contains large solid islands of lobular carcinoma cells. It is an important variant to recognize histologically as it can be mistaken for lymphoma.
5.3.2 Alveolar Lobular Carcinoma
The histology of the alveolar variant of lobular carcinoma shows loose sheets and nests of carcinoma cells with central spaces or ‘alveoli.’ These are low-grade (Grade 1) tumors.
Figure 5.13 Solid and Alveolar Variants
of Lobular Carcinoma
Photomicrographs of the histology of A. solid invasive lobular carcinoma
(ILC) with sheets of non-cohesive small cells with little pale cytoplasm;
B. alveolar pattern of invasive lobular carcinoma (ILC) with cells
surrounding spaces, some filled with secretions. (H&E x40)
5.3.3 Invasive Pleomorphic Lobular Carcinoma (IPLC)
Invasive pleomorphic lobular carcinoma (IPLC) has recently been defined as a carcinoma with a lobular growth pattern and marked nuclear pleomorphism (pleomorphism 3) (Rakha et al., 2013). Histological grade (in particular the mitotic cell count) in ILC has been shown to be of prognostic importance. IPLC is now considered as a unique sub-type of breast cancer.
IPLC is not an easy breast cancer to diagnose, or rather, to classify. ‘Pleomorphic’ cancer cells can occur in other kinds of breast cancer, such as sarcomas of the breast and metastatic tumors to the breast.
To make a classification even more difficult, ‘spindle cell’ differentiation may be found in 40% of cases and can make up more than 25% of the tumor volume.
Unlike the more common variants of invasive lobular carcinoma (ILC), the pleomorphic variant can present as a palpable breast mass, with a minority found on screening mammography.
Mammograms of pleomorphic ILC will tend to show well-circumscribed lesions without evidence of calcification.
Sonograms of pleomorphic ILC tend to show hypoechoic tumors or tumors with heterogeneous internal echoes.
Magnetic resonance imaging (MRI) can be used to diagnose pleomorphic lobular breast carcinoma; it will tend to show a homogeneous mass that might contain some small cysts.
Pleomorphic ILC is a Grade 3 tumor with abundant mitotic figures. These cancers are negative for estrogen receptors (ER); most are also negative for progesterone receptors (PR).
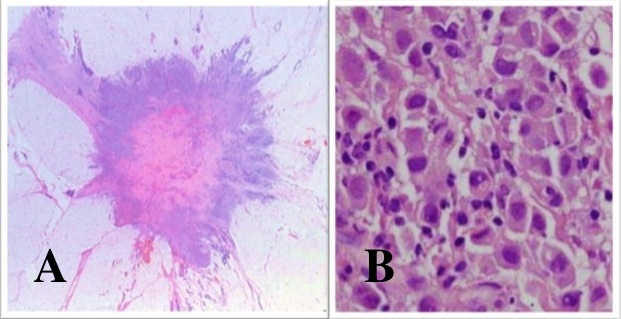
Figure 5.14 Invasive Pleomorphic Lobular Carcinoma (IPLC).
A. Low power scanning photomicrograph of the pleomorphic variant
of invasive lobular carcinoma (IPLC). B. High power photomicrograph
shows non-cohesive, small cells of various shapes and size. (H&E x40)
5.3.4 Tubulo-Lobular Carcinoma
Tubulo-lobular or ‘tubular-lobular’ carcinoma is a breast cancer variant that has hybrid histologic characteristics of invasive tubular carcinoma and invasive lobular carcinoma. The tumor cells are arranged in well-formed glands and single-file infiltrates.
The radiologic appearances are similar to invasive lobular carcinoma, and there is the same tendency to multifocality and multi-centricity.
In terms of staging, these tumors behave more like invasive, moderately-differentiated ductal carcinoma. Often these tumors are ‘misclassified’ as invasive carcinoma with mixed ductal and lobular features (Rakha & Ellis, 2010).
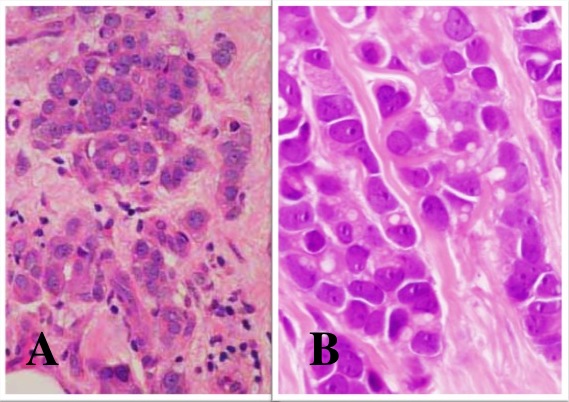
Figure 5.15 Tubulo-Lobular Carcinoma
Photomicrographs of the histology show A. tubule formation with some
single-cell infiltrates; B. higher power image shows the cell morphology is
typical for lobular carcinoma, with eccentric cell nuclei. (H&E x40 x60)
5.3.5 Mixed Lobular Carcinoma
On histology, the mixed variant of lobular carcinoma can contain a variety of the appearances described and in varying proportions (Rakha & Ellis, 2010).
5.4 Tubular Carcinoma
Tubular breast cancer has become more commonly diagnosed with the advent of mammographic breast screening, but these are still quite rare breast cancers.
Tubular carcinomas are usually about 1 cm in diameter or less; they are usually low-grade. The average age for diagnosis of tubular carcinoma is around the early 50’s. However, tubular carcinoma can occur in women at any age.
Tubular carcinoma has a favorable prognosis with low-grade tumor cell nuclear features.
2012 WHO guidelines recommend strict diagnostic requirements for tubular carcinoma, with characteristic features being present in > 90 % of the tumor, when examined microscopically.
On microscopy, tubular carcinoma consists of well-formed tubular or glandular structures infiltrating the breast stroma; these tubules are elongated with pointed ends. The cells are cuboidal to columnar, with apical cytoplasmic protrusions or ‘snouts.’
Tubular carcinoma is a low-grade carcinoma, with a relatively good prognosis. The presence of low-grade DCIS is present in about 60% of cases. Metastases of tubular carcinoma of the breast are rare (Sullivan et al., 2005).
Figure 5.16 Tubular Carcinoma
A. Low power scanning photomicrograph of the infiltrating edge of a
tubular carcinoma shows irregular tubules with pointed ends,
randomly distributed in fibrotic breast stroma. (H&E x20)
B. High power photomicrograph of the ‘tubules’ shows atypical cells
with protrusions or cell ‘snouts.’ (H&E x 60)
5.5 Cribriform Carcinoma
Invasive cribriform carcinoma also has a favorable prognosis. Indeed, 2012 WHO guidelines recommend strict diagnostic requirements, with characteristic features being present in > 90% of the tumor.
On microscopy, invasive cribriform carcinoma shows packets of cells with lattice-like formations, invading the breast tissue.
Invasive cribriform carcinoma is usually of low-grade. Usually, ductal carcinoma in-situ (DCIS) of the cribriform type is also present.
A cribriform pattern can be seen in about 5% of invasive ductal carcinoma (IDC) of the breast.
Figure 5.17 Invasive Cribriform Carcinoma
Photomicrograph of the invasive cancer cells that form
lattice-like groups. (H&E x 20)
5.6 Mucinous (Colloid) Carcinoma
Mucinous or ‘colloid’ carcinoma accounts for between 1% and 2% of invasive breast cancers. In pure form, mucinous carcinoma has a good prognosis (Type A subtype). But a hypercellular or large cell form (Type B) is now shown to have a prognosis similar to that of NST tumors.
The SEER data analysis show that mucinous carcinoma cases have the oldest mean age at diagnosis (around 65 years).
Mucinous carcinoma has a soft, gelatinous appearance on gross examination, and they tend to be well-circumscribed.
Mucinous carcinoma is characterized microscopically by nests of tumor cells within ‘pools’ of extracellular mucin. The tumor cells have a uniform, low-grade cell nuclei.
As with tubular carcinoma and cribriform carcinoma, mucinous carcinoma has a more favorable prognosis.
Figure 5.18 Mucinous ‘Colloid’ Carcinoma
Photomicrograph of A. small groups of tumor cells ‘floating’
in ‘pools’ of mucin; B. higher power view of the cells show atypical
cytology but cohesive cells are seen. (H&E x 4 and x 60)
5.7 Carcinoma with Medullary Features
‘Carcinoma with medullary features’ is the name now recommended by 2012 WHO guidelines as the term for tumors formerly known as ‘medullary carcinoma‘,‘ ‘atypical medullary carcinoma’ and ‘invasive carcinoma NST with medullary features.’
Medullary carcinoma accounts for between 1% to 10 % of invasive breast cancers.
Carcinoma with medullary features occurs more frequently in younger patients than other types of breast cancer. They are more frequent in women with mutations of the BRCA1 gene (10 % of breast cancers are medullary in this population, as compared with <1 % of non-BRCA1–related breast cancers). However, the majority of breast cancers in patients with BRCA1 gene mutations (90 %) are not medullary
Mammographic X-ray shows a rounded or ovoid mass with ill-defined margins. Micro-calcification is not a feature.
On gross examination, medullary carcinoma is well-circumscribed, soft and tan-brown with areas of hemorrhage or necrosis.
On microscopy, the tumor cells appear to be poorly-differentiated (high-grade), the cells grow in a syncytial pattern (they appear to fuse), and have an intense associated lymphoplasmacytic infiltrate.
This group of tumors has a relatively good prognosis, possibly due to the associated lymphocyte infiltrate and their presence in patients with BRCA1 germline mutations.
The prognosis for pure medullary carcinoma is more favorable than that of infiltrating ductal carcinoma (IDC), despite the tumor’s appearance.
Figure 5.19 Carcinoma with Medullary Features
Photomicrograph of the histology of medullary carcinoma shows
large, pale cells with indistinct cell borders, giving the appearance of
‘syncytial’ formations; there are lymphocytes encircling the tumor.
(H&E x 40)
5.8 Carcinoma with Apocrine Differentiation
The most common category for carcinoma with apocrine differentiation is of ‘no specific type’ (NST)
Apocrine differentiation occurs in ‘special-type’ carcinomas, such as lobular, papillary, and medullary tumors. For this reason, the use of the term ‘apocrine carcinoma’ is now discouraged.
The WHO (2012) now proposes that tumors with apocrine differentiation are classified according to their primary invasive component (ductal or lobular).
Figure 5.20 Breast Carcinoma with Apocrine Differentiation
Photomicrograph of the histology from an invasive ductal carcinoma of the
breast that contains foci of apocrine cell differentiation, such as this; this
cancer would be called an ‘invasive ductal carcinoma with
apocrine differentiation.’ (H&E x 100)
5.9 Carcinoma with Signet-Ring Cell Differentiation
The 2012 WHO classification, groups together signet ring cell carcinoma with mucinous carcinoma. So, signet cell carcinoma is no longer a separate condition of the breast.
Signet-ring cell differentiation can be present in several tumor types, including invasive lobular carcinomas (ILC) or with invasive carcinomas NST, and with high-grade mucinous carcinomas.
Signet-ring differentiation does not have any prognostic significance.
Figure 5.21 Breast Carcinoma with Signet-Ring Cell Differentiation
Photomicrograph of the histology of an invasive lobular carcinoma shows foci of signet-ring differentiation; this cancer would be called an ‘invasive lobular carcinoma with signet-ring differentiation.’ (H&E x 20)
Further Reading
- Types of Invasive Lobular Carcinoma
- Lobular Carcinoma In-Situ and Invasive Lobular Carcinoma
- Ductal Carcinoma In-Situ and Lobular Carcinoma In-Situ
- Lobular Carcinoma In-Situ (LCIS)
- Index of ALL our Articles on Types of Breast Cancer
- Index of ALL our Articles on Breast Cancer Incidence and Survival Rates
- Articles about Breast Cancer Treatment
References
Li, C.I., Anderson, B.O., Daling, J.R., Moe, R.E. (2003). Trends in incidence rates of invasive lobular and ductal breast carcinoma. JAMA 289, 1421–1424. (Retrieved November 13th 2014): https://www.ncbi.nlm.nih.gov/pubmed/12636465
Rakha, E.A., van Deurzen, C.H., Paish, E.C., Macmillan, R.D., Ellis, I.O., Lee, A.H. (2013). Pleomorphic lobular carcinoma of the breast: is it a prognostically significant pathological subtype independent of histological grade? Mod Pathol 26(4), 496-501. (Retrieved November 12th 2014): https://www.ncbi.nlm.nih.gov/pubmed/23238630
Patient Information
Breast Cancer Org.ILC – Invasive Lobular Cancer. (Retrieved February 13th 2015): http://www.breastcancer.org/symptoms/types/ilc
Surveillance, Epidemiology, and End Results Program (SEER) (Retrieved February 23rd 2015): http://seer.cancer.gov/
Forward to 5C on papillary types Back to 5A ductal cancer types


